An FDA approved CLIA Waived rapid point-of-care test that detects antibodies to HIV-1 and HIV-2 in fingerstick whole blood, venous whole blood, serum and plasma specimens.
*Refer to product insert for more details
Test for HIV in 3 Easy Steps with SURE CHECK®
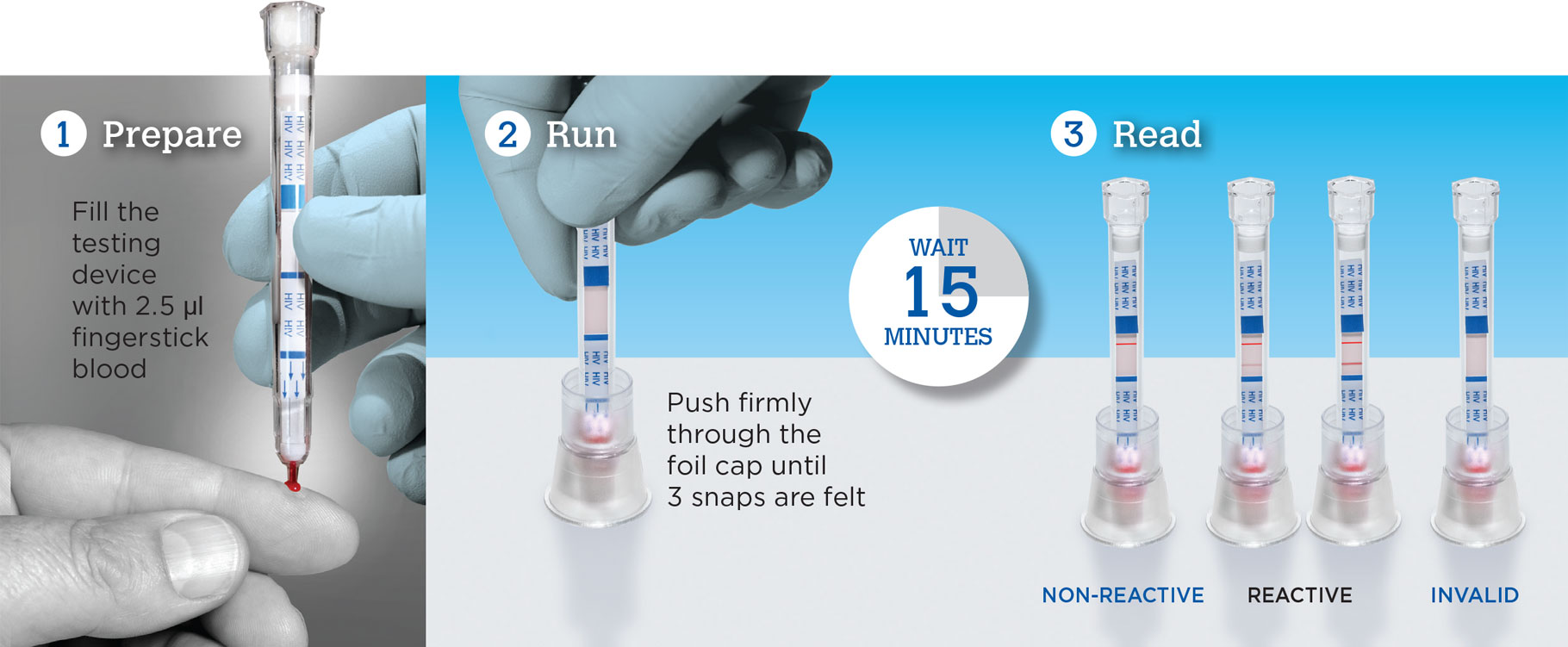
SURE CHECK HIV Test: Rapid HIV Test in 3 Easy Steps*
This video will show you how to use Chembio’s SURE CHECK HIV 1/2 Assay.
Product Information
| Information Type | Product Detail | |
| Sensitivity | 99.7% | |
| Specificity | 99.9% | |
| Method | Lateral Flow | |
| Time to Results | 15 minutes | |
| Test and External Controls Shelf Life** | 24 months | |
| Storage Conditions | 8 - 30°C (46-86°F) | |
**From date of manufacture.
Product Downloads & Links
Please fill out the form below to receive the following downloads and links:
- Product Insert
- Quick Reference Guide
- Product MSDS
- Control Pack Insert
- Control Pack MSDS
Ordering Information
| Product | Catalog Number | |
| SURE CHECK® HIV 1/2 Assay (25 Tests) FDA Approved, CLIA Waived | 60-9507-0 | |
| HIV Rapid Test Control Pack | 60-9549-0 | |
Reimbursement Codes: 86703 or 86703QW
G0435 (for Medicare)
Reimbursement Codes: 86703 or 86703QW
G0435 (for Medicare)
More Reimbursement Info