Contact your local Chembio Representative to learn more about our Detect test.
An FDA EUA visual read rapid test for detection diagnosis of SARS-CoV-2 Antigen in nasal swab specimens.
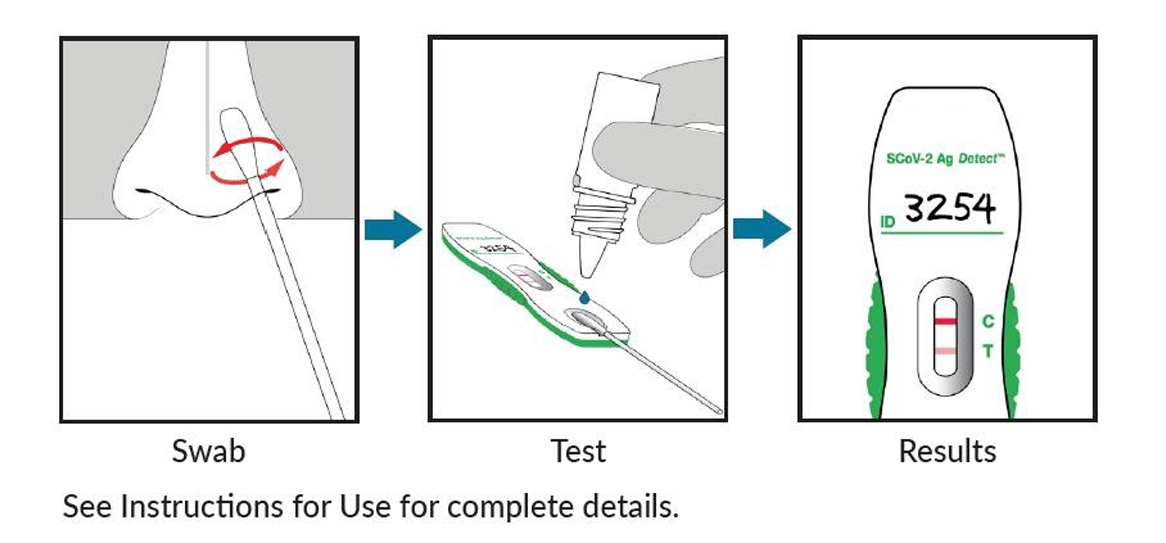
Test for SARS-CoV-2 in 3 Easy Steps with SCoV-2 DetectTM*
Product Information*
| Information Type | Product Details |
| Time to Results | 20 Minutes |
| Sample | Nasal Swab Specimen |
| Reader | No analyzer required; Visual Read |
| Storage Conditions | 59° to 86°F (15° to 30°C) |
| Reimbursement | COVID-19: 87811-QW More Reimbursement Info |
| Method | Lateral Flow |
Ordering Information
| Product | # of Tests | Catalog Number |
| SCoV-2 Detect™ | 50 | COVAG-RC-50 |
| SCoV-2 Detect™ | 20 | COVAG-20-20 |
*This Test has not been FDA cleared or approved but has been authorized by FDA under an EUA for use by authorized laboratories. It has been authorized only for the detection of proteins from SARS-CoV-2, not for any other viruses or pathogens. Its emergency use is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostics for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Federal Food, Drug and Cosmetic Act, 21 U.S.C. § 360bbb-3(b)(1), unless the declaration is terminated or authorization is revoked sooner.